Johnson & Johnson is stepping into the robotic surgery arena with their new OTTAVA™ system. It’s a big move for the company, aiming to bring something new to minimally invasive procedures. They’ve been working on this for a while, and it looks like they’re ready to show what it can do. This system is designed to be flexible and grow with future technology, which is pretty interesting.
Key Takeaways
- The JNJ robotic surgery OTTAVA™ system is designed to handle a variety of general surgery procedures in the upper abdomen.
- Johnson & Johnson has submitted the OTTAVA™ system for De Novo classification with the FDA, using data from its investigational device exemption (IDE) study.
- A second IDE study is approved for the OTTAVA™ system in inguinal hernia procedures, a common surgery in the U.S.
- The OTTAVA™ system features a unified architecture, Ethicon surgical instruments, and plans to integrate with the Polyphonic™ digital ecosystem.
- JNJ robotic surgery aims to compete in a market currently dominated by other players, offering more choices for surgeons and hospitals.
Introducing the JNJ Robotic Surgery OTTAVA™ System
A New Era in Minimally Invasive Procedures
Johnson & Johnson is stepping into the future of surgery with its new OTTAVA™ Robotic Surgical System. This isn’t just another piece of equipment; it’s designed to change how surgeons approach minimally invasive procedures. Think about it – operations that used to require big cuts can now be done with much smaller ones, leading to quicker recovery times for patients. The OTTAVA system aims to make these advanced techniques more accessible and effective. It’s built on years of experience in surgery and a deep understanding of what surgeons need in the operating room.
Addressing Unmet Needs in Robotic Surgery
For a while now, robotic surgery has been around, but there have been some persistent challenges. Surgeons have pointed out areas where current systems just don’t quite measure up. JNJ’s OTTAVA system was developed with these specific issues in mind. It’s not just about having a robot; it’s about having a robot that works better for a wider range of surgeries and patient anatomies. The goal is to fill those gaps and provide tools that truly make a difference in patient care.
Designed for Versatility and Future Growth
One of the really interesting things about OTTAVA is how it’s built. It has a unified architecture, which basically means all the parts work together really smoothly. This design isn’t just for today; it’s made to be adaptable. Johnson & Johnson is thinking ahead, planning for how this system can grow and connect with other digital tools in the future, like the Polyphonic™ digital ecosystem. This means it can handle a lot of different types of surgeries now and be updated for new procedures and technologies down the line. It’s like building a foundation that can support many different kinds of buildings later on.
Clinical Validation and Regulatory Milestones
Investigational Device Exemption Study Success
The journey of the OTTAVA™ system to market has been marked by careful testing and regulatory steps. A key part of this was the Investigational Device Exemption (IDE) study. This study allowed the system to be used in real-world clinical settings to gather important data. The initial focus was on Roux-en-Y gastric bypass procedures, a significant surgery for weight management. The results from this study were really important, providing the evidence needed for the next big step.
De Novo Classification Submission
With the successful completion of the IDE study, Johnson & Johnson took the OTTAVA™ system to the U.S. Food and Drug Administration (FDA). They submitted an application seeking a De Novo classification. This is a special pathway for novel medical devices that don’t have a direct equivalent already on the market. Getting this classification means the FDA has reviewed the device and found it to be low-to-moderate risk, clearing the way for marketing authorization. The submission specifically targeted multiple general surgery procedures in the upper abdomen, showing the system’s intended broad application.
Expanding Trial Scope to Hernia Repair
But the work didn’t stop there. Johnson & Johnson also secured approval for another IDE, this time to begin a U.S. clinical trial for the OTTAVA™ system in inguinal hernia repair. Hernia repair is one of the most common surgeries performed today, so expanding the trial scope to include this procedure highlights the system’s versatility. This next phase of testing will gather more data, further supporting the system’s safety and effectiveness across a wider range of surgical needs.
Technological Innovations of the OTTAVA™ System
Unified Architecture for Enhanced Performance
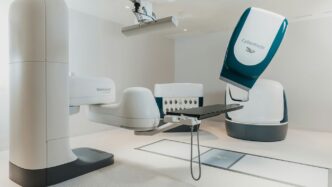
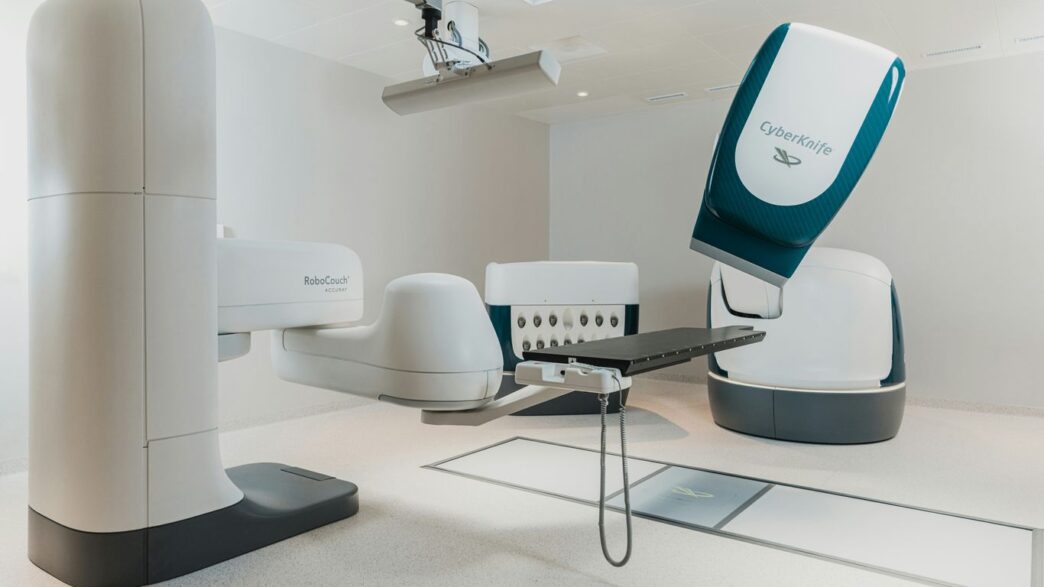
The OTTAVA™ system is built around a really smart, unified design. Think of it like a single, cohesive unit rather than a bunch of separate parts trying to work together. This approach means everything is more streamlined, which should lead to better performance during surgery. It’s designed to be compact, with robotic arms that tuck away neatly under the operating table. What’s neat is that the table and the arms can move together during an operation, allowing surgeons to adjust their view and access without a lot of fuss. This flexibility is a big deal for complex procedures.
Ethicon Expertise in Surgical Instrumentation
Johnson & Johnson isn’t new to surgery, and they’re bringing all that experience to OTTAVA, especially with their Ethicon instruments. These are the tools surgeons use, and the ones designed for OTTAVA are made to work smoothly with the robotic system. The idea is that the feel and consistency of these instruments will be familiar to surgeons, whether they’re using the robot or traditional laparoscopic tools. This connection between the robotic system and trusted surgical instruments is key to making the transition easier for surgical teams.
Integration with the Polyphonic™ Digital Ecosystem
Looking ahead, OTTAVA is set up to connect with Johnson & Johnson’s Polyphonic™ digital platform. This isn’t just about the robot itself; it’s about creating a connected surgical environment. Polyphonic aims to link different surgical technologies, robotics, and software. The goal is to gather data and provide insights that can help surgeons make better decisions during operations. This digital integration is what really sets OTTAVA up for the future, making surgery smarter and more informed.
JNJ Robotic Surgery: A Competitive Landscape
Challenging Market Dominance
The world of robotic surgery has been pretty much a one-horse race for a long time, with Intuitive Surgical’s da Vinci system being the go-to choice for many. But Johnson & Johnson is stepping into the ring with its OTTAVA™ system, aiming to shake things up. They’re not just looking to offer another option; they’re trying to address things that surgeons have found lacking in current systems. This move signals a significant shift, promising more choices for hospitals and surgeons.
Driving Choice and Competition
It’s no secret that having more players in a market usually means better innovation and, hopefully, better prices. JNJ is betting that by bringing their extensive experience in surgical tools and medical devices to the table, they can create a system that really meets the needs of modern surgery. They’ve talked about how OTTAVA is designed to be adaptable and ready for future tech, which is a smart play in a fast-moving field. It’s about making sure surgeons have the best tools available, not just the most established ones.
Leveraging Johnson & Johnson’s Surgical Legacy
Johnson & Johnson has been around the block in surgery for over a century. They’ve got a deep history with things like wound closure, surgical staplers, and instruments through their Ethicon brand. Now, they’re combining all that know-how with robotics. The OTTAVA system is built on this foundation, aiming to integrate smoothly with existing surgical practices while introducing new robotic capabilities. They’re also connecting it to their Polyphonic™ digital platform, which sounds like it could help surgeons make better decisions by pulling together data from different sources. It’s a big push to bring their long-standing surgical credibility into the robotic era.
The Future of JNJ Robotic Surgery
Potential Impact on General Surgery
The OTTAVA™ system is really shaping up to be a game-changer, especially for general surgery. Think about it, procedures that used to require big incisions or a whole team of surgeons might become much more manageable. JNJ is aiming to make things like gastric bypass and hernia repairs more accessible and maybe even quicker for patients. They’ve submitted for de novo classification, which is a big step, showing they’re serious about getting this into hospitals.
Enabling Complex Multi-Quadrant Procedures
What’s really interesting is how OTTAVA™ is designed for those really tricky surgeries. You know, the ones where the surgeon needs to reach different parts of the abdomen, sometimes called multi-quadrant procedures. The system’s setup, with arms integrated into the table that can move around, seems like it could make these complex operations less of a juggling act. It’s not just about doing simple things better; it’s about making the really hard stuff more doable.
Commitment to Advancing Surgical Care
Johnson & Johnson has a long history in surgery, and this feels like the next big step. They’re not just putting out a new robot; they’re talking about a whole digital ecosystem, called Polyphonic™, that connects everything. This means more data and insights for surgeons, which could lead to better decisions during operations. It’s clear JNJ sees robotic surgery as a key part of the future, and they’re investing heavily to push that forward. They’re looking to bring more options to the market, which is good news for hospitals and ultimately, for patients.
Wrapping Things Up
So, it looks like Johnson & Johnson is really pushing forward with their OTTAVA robotic surgery system. They’ve submitted it to the FDA, which is a big step, and they’re aiming for it to be used in a bunch of different surgeries, especially in the upper abdomen. They’ve also got another trial going for hernia repairs. It’s clear they want to shake things up in the robotic surgery world, which has been pretty dominated by one company for a long time. This system is designed to be flexible and connect with other tech down the line. It’ll be interesting to see how it all plays out and if it really makes a difference for surgeons and patients.